Chemical Equations and Reactions Chapter 8 Review Answer Key

Chemical Equations and Reactions Chapter viii

Describing Chemic Reactions Section ane

Chemic Reactions • A chemic equation represents, with symbols and formulas, the identities and relative molecular or tooth amounts of the reactants and products in a chemic reaction. – example: The post-obit chemical equation shows that the reactant ammonium dichromate yields the products nitrogen, chromium(Three) oxide, and water. (NH 4)2 Cr 2 O vii(due south) N 2(thousand) + Cr two O 3(s) + 4 H 2 O(g)

Indications of a Chemical Reaction • Certain easily observed changes ordinarily indicate that a chemical reaction has occurred. 1. Development of free energy every bit heat and light - Magnesium example 2. Product of a gas - Bubbles – Baking soda and vinegar – CO two 3. Formation of a precipitate. • A solid that is produced as a result of a chemic reaction in solution and that separates from the solution is known as a precipitate. 4. Color change

Characteristics of Chemical Equations • The following requirements will aid you in writing and reading chemical equations correctly. 1. The equation must represent known facts. two. The equation must incorporate the right formulas for the reactants and products. 3. The police force of conservation of mass must be satisfied. • A coefficient is a small whole number that appears in front of a formula in a chemical equation.

Elements That Normally Be as Diatomic Molecules

Word and Formula Equations • The first step in writing a chemic equation is to identify the facts to exist represented. • A give-and-take equation is an equation in which the reactants and products in a chemical reaction are represented by words. – A discussion equation is qualitative • example: methane + oxygen carbon dioxide + water

Word and Formula Equations • The next step in writing a correct chemical equation is to replace the names of the reactants and products with appropriate symbols and formulas. • A formula equation represents the reactants and products of a chemical reaction by their symbols or formulas. • example: The formula equation for the reaction of methyl hydride and oxygen is: CH four(grand) + O ii(m) CO 2(thou) + H two O(chiliad) (not balanced)

Word and Formula Equations • To complete the procedure of writing a right equation, the constabulary of conservation of mass must exist taken into account. • The relative amounts of reactants and products represented in the equation must exist adjusted and so that the numbers and types of atoms are the aforementioned on both sides of the equation. • This procedure is called balancing an equation and is carried out past inserting coefficients.

Word and Formula Equations • To balance the equation, begin by counting atoms of elements that are combined with atoms of other elements and that appear only once on each side of the equation. CH 4(grand) + O 2(g) CO 2(g) + 2 H ii O(g) (not balanced) • Begin by counting carbon atoms. • Carbon is already balanced in the equation. • 2 additional hydrogen atoms are needed on the right side of the equation.

Word and Formula Equations CH iv(g) + O 2(1000) CO 2(g) + ii H 2 O(g) (partially balanced) • Now consider the number of oxygen atoms. • Increase the number of oxygen atoms on the left side to 4 past placing the coefficient ii in front end of the molecular formula for oxygen. • The right chemical equation, or counterbalanced formula equation, for the burning of methane in oxygen is CH 4(one thousand) + 2 O 2(1000) CO two(k) + 2 H ii O(g)

Symbols Used in Chemical Equations

Symbols Used in Chemical Equations

Symbols Used in Chemic Equations

Methyl hydride Combustion

Example • Write give-and-take and formula equations for the chemical reaction that occurs when solid sodium oxide is added to water at room temperature and forms sodium hydroxide (dissolved in the water). Include symbols for physical states in the formula equation. Then balance the formula equation to give a balanced chemical equation.
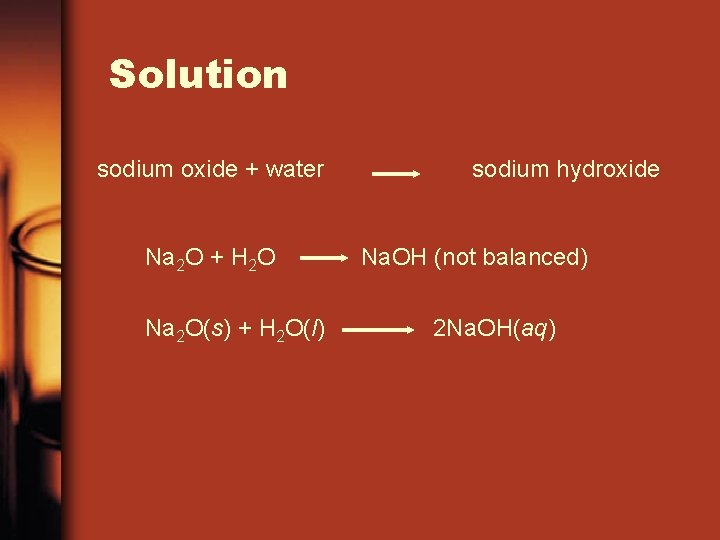
Solution sodium oxide + water Na 2 O + H 2 O Na ii O(s) + H two O(l) sodium hydroxide Na. OH (not balanced) 2 Na. OH(aq)
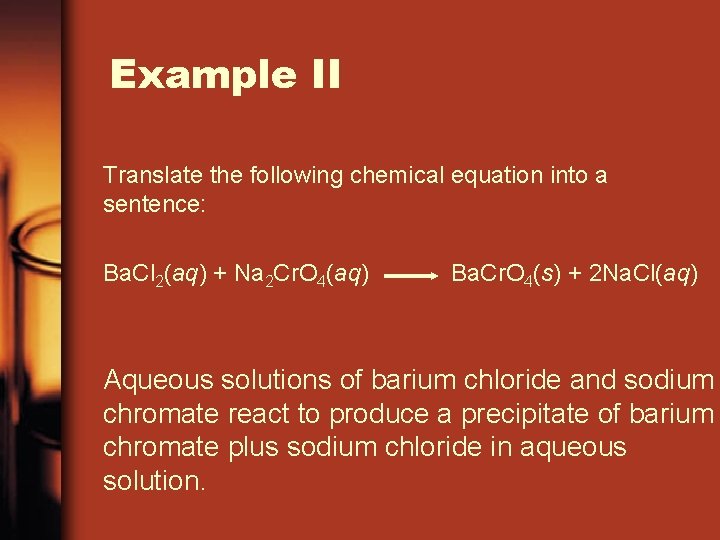
Example II Interpret the post-obit chemical equation into a sentence: Ba. Cl two(aq) + Na ii Cr. O 4(aq) Ba. Cr. O four(south) + 2 Na. Cl(aq) Aqueous solutions of barium chloride and sodium chromate react to produce a precipitate of barium chromate plus sodium chloride in aqueous solution.

Your Plow I Translate these Equations into sentences: CS two (l) + 3 O 2 (g) CO 2 (chiliad) + two And so 2 (g) Copper disulfide and oxygen gas react to produce carbon dioxide gas and sulfur dioxide gas Na. Cl (aq) + Ag. NO 3 Na. NO 3 (aq) + Ag. Cl (south) Aqueous sodium chloride and silverish nitrate react to produce an aqueous solution of sodium nitrate and a precipitate of silverish chloride
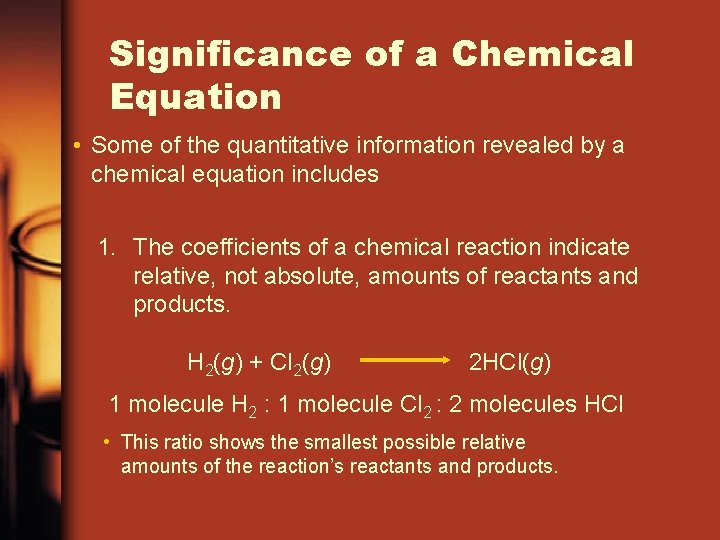
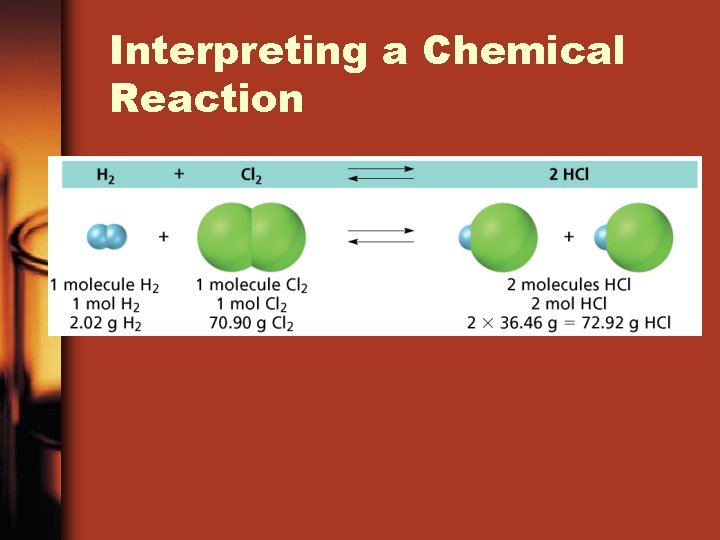
Significance of a Chemical Equation • Some of the quantitative information revealed by a chemical equation includes ane. The coefficients of a chemical reaction indicate relative, not absolute, amounts of reactants and products. H two(g) + Cl 2(g) 2 HCl(g) ane molecule H 2 : ane molecule Cl 2 : 2 molecules HCl • This ratio shows the smallest possible relative amounts of the reaction's reactants and products.

Significance of a Chemical Equation ii. The relative masses of the reactants and products of a chemical reaction can exist determined from the reaction's coefficients. • An amount of an chemical element or compound in moles tin exist converted to a mass in grams past multiplying past the appropriate molar mass. • example:
Interpreting a Chemical Reaction

Significance of a Chemical Equation 3. The reverse reaction for a chemical equation has the aforementioned relative amounts of substances as the forward reaction. • An equation gives no indication of whether a reaction will really occur. • Chemic equations give no information most the speed at which reactions occur. • Equations do not give any data near how the bonding between atoms or ions changes during the reaction.

Visual Concept • Equation

Balancing Chemical Equations • The following procedure demonstrates how to chief balancing equations by inspection using a step-bystep approach. 1. Place the names of the reactants and the products, and write a give-and-take equation. h2o hydrogen + oxygen

Balancing Chemical Equations 2. Write a formula equation by substituting right formulas for the names of the reactants and the products. H 2 O(50) H two(chiliad) + O ii(one thousand) (non balanced)

Balancing Chemical Equations 3. Balance the formula equation according to the police force of conservation of mass. • Residuum the different types of atoms one at a time. • First remainder the atoms of elements that are combined and that announced only once on each side of the equation. • Rest polyatomic ions that appear on both sides of the equation as single units. • Balance H atoms and O atoms after atoms of all other elements have been counterbalanced.

Balancing Chemical Equations iii. Rest the formula equation according to the law of conservation of mass. • Residual oxygen atoms past increasing the number of H two O molecules. ii H 2 O(l) H 2(one thousand) + O two(grand) (partially balanced) • Residue the hydrogen atoms by placing the coefficient two in forepart of hydrogen, H 2. two H 2 O(l) 2 H 2(g) + O 2(yard) (balanced)

Balancing Chemical Equations 4. Count atoms to exist sure that the equation is balanced. 2 H ii O(l) • two H ii(1000) + O two(chiliad) H 4 O two If the coefficients do not represent the smallest possible whole-number ratio of reactants and products, divide the coefficients by their greatest common factor in gild to obtain the smallest possible whole-number coefficients.

Never • Change a subscript to balance an equation. • If you lot change the formula you are describing a dissimilar reaction. • H ii O is a dissimilar compound than H 2 O 2 • Never put a coefficient in the eye of a formula • 2 Na. Cl is okay, Na two Cl is not.

Visual Concept • Balancing Equations

Example • The reaction of zinc with aqueous hydrochloric acrid produces a solution of zinc chloride and hydrogen gas. Write a balanced chemical equation for the reaction.

Solution • Write the discussion equation. zinc + hydrochloric acid zinc chloride + hydrogen • Write the formula equation. Zn(due south) + HCl(aq) Zn. Cl 2(aq) + H 2(thou) (not balanced) • Adjust the coefficients. • Balance chlorine showtime considering it is combined on both sides of the equation. Zn(s) + 2 HCl(aq) Zn. Cl two(aq) + H 2(g) • Count atoms to check balance. Zn(due south) + 2 HCl(aq) Zn. Cl 2(aq) + H 2(g) (ane Zn) + (2 H + 2 Cl) = (one Zn + two Cl) + (2 H)

Your Turn II a) Lead(NO iii)2 + K 2 Cr. O 4 Pb. Cr. O iv + KNO three b) Mn. O 2 + HCl Mn. Cl 2 + H ii O+ Cl two c) C 3 H 6 + O 2 CO 2 +H 2 O d) Zn(OH)2 + H 3 PO 4 Zn 3(PO 4)2 + H ii O e) CO + Iron two O iii Fe + CO 2 f) CS 2 + Cl 2 CCl 4 +S 2 Cl 2 g) CH 4 + Br 2 CH iii Br + HBr h) Ba(CN)two + H 2 And so 4 Ba. So 4 + HCN

Answers a) Pb(NO iii)2 + One thousand 2 Cr. O 4 Pb. Cr. O 4 + two KNO 3 b) Mn. O 2 + 4 HCl Mn. Cl 2 + 2 H 2 O+ Cl 2 c) 2 C 3 H 6 + nine O 2 6 CO two +6 H 2 O d) three Zn(OH)2 + 2 H 3 PO 4 Zn 3(PO 4)2 + 6 H ii O east) iii CO + Fe 2 O three two Atomic number 26 + 3 CO 2 f) CS 2 + 3 Cl 2 CCl 4 +S 2 Cl two g) CH 4 + Br two CH 3 Br + HBr h) Ba(CN)two + H 2 So 4 Ba. And so iv + 2 HCN

Ms Houts' Tricks • If an atom appears more than than once on a side, balance it terminal. • If you fix everything except one element, and it is even on one side and odd on the other, double the first number, and then move on from at that place. C four H 10 + O 2 CO 2 + H 2 O

Types of Chemical Rx. Due north'southward Section two

Types of Rx. N's • • • There are millions of reactions. Can't call back them all Autumn into several categories. We volition larn 5 types. Volition be able to predict the products. For some we will exist able to predict whether they will happen at all. • Volition recognize them by the reactants

Types of Rx. N's • The nomenclature scheme described in this department provides an introduction to 5 basic types of reactions: • Synthesis (Combination) • Decomposition • Single-displacement • Double-displacement • Combustion reactions

Synthesis Reactions • In a synthesis reaction, also known as a limerick reaction or combination reaction, two or more substances combine to course a new compound. • This type of reaction is represented by the following general equation. A+10 AX • A and X tin can be elements or compounds. • AX is a compound

Synthesis Reactions • 2 elements, or compounds combine to make 1 compound. • Ca +O 2 Ca. O • And so 3 + H 2 O H 2 So 4 • We tin predict the products if they are two elements. • Mg + N 2

Reactions of Elements with Oxygen • 1 simple type of synthesis reaction is the combination of an element with oxygen to produce an oxide of the element. • Virtually all metals react with oxygen to form oxides. • example: ii Mg(s) + O 2(m) 2 Mg. O(s) • Group ii elements react in a similar manner, forming oxides with the formula MO, where M represents the metallic.

Your Plough Iii Ca + Cl 2 Fe + O two iron (II) oxide Al + O 2 Call back that the starting time footstep is to write the formula • Then balance • •

Decomposition Reactions • In a decomposition reaction, a single compound undergoes a reaction that produces 2 or more simpler substances. s or compounds. • Decomposition reactions are the opposite of synthesis reactions. • They are represented by the following general equation. AX A+X • AX is a compound. • A and Ten can be elements or compounds.

Decomposition of Binary Compounds • The decomposition of a substance past an electric current is called electrolysis. • case: • Oxides of the less-active metals, which are located in the lower middle of the periodic table, decompose into their elements when heated. • example:

Decomposition Reactions Decomposition of Metal Carbonates Decomposition of Metal Hydroxides Decomposition of Metal Chlorates

Your Plough Four • Ni. CO 3 • H ii CO 3(aq)

Single-Replacement Reactions • In a unmarried-replacement reaction, also known as a deportation reaction, one chemical element replaces a like element in a compound. • Many single-replacement reactions take place in aqueous solution. • Single-replacement reactions tin can be represented by the following general equations. A + BX AX + B or Y + BX By + X • A, B, 10, and Y are elements. AX, BX, and BY are compounds.

Unmarried-Replacement Reactions • 1 element replaces another • Reactants must exist an element and a compound. • Products will be a dissimilar chemical element and a unlike compound. • Na + KCl K + Na. Cl • F 2 + Li. Cl Li. F + Cl ii

Single-Replacement Reactions • • • Metals supersede metals (and hydrogen) M + Al. N Zn + HCl Think of water as HOH Metals supersede one of the H, combine with hydroxide. • Na + HOH

Activity Serial • We tin tell whether a reaction will happen • Some are more active than other • More agile replaces less agile • There is a list on page 286 • Higher on the listing replaces lower. • If the chemical element by itself is higher, it happens, if lower information technology doesn't

Reactivity Series

Activity Series • H can be replaced in acids by everything higher • Just the first vi (Li - Na) react with water. • Fe + Cu. SO 4 • Lead + KCl • Al + HCl

Activeness Serial • What does information technology mean that Au And Ag are on the bottom of the list? • Nonmetals tin can supervene upon other nonmetals • Limited to F 2 , Cl 2 , Br two , I ii • The order of activity is that on the tabular array. • Higher replaces lower. • F 2 + HCl • Br two + KCl

Double-Replacement Reactions • In double-replacement reactions, the ions of two compounds commutation places in an aqueous solution to form ii new compounds. • One of the compounds formed is usually a precipitate, an insoluble gas that bubbling out of the solution, or a molecular compound, normally water. • The other compound is often soluble and remains dissolved in solution.

Double-Replacement Reactions • A double-displacement reaction is represented by the following general equation. AX + BY AY + BX • A, X, B, and Y in the reactants correspond ions. • AY and BX represent ionic or molecular compounds.

Double-Replacement Reactions • The formation of a precipitate occurs when the cations of 1 reactant combine with the anions of some other reactant to grade an insoluble or slightly soluble compound. • case: 2 KI(aq) + Pb(NO 3)2(aq) Lead. I 2(due south) + ii KNO iii(aq) • The precipitate forms as a result of the very potent bonny forces between the Lead two+ cations and the I anions.

Double-Replacement Reactions • Two things replace each other. • Reactants must exist two ionic compounds or acids. • Normally in aqueous solution • Na. OH + Fe. Cl iii • The positive ions alter place. • Na. OH + Fe. Cl 3 Iron+3 OH- + Na+1 Cl-1 • Na. OH + Iron. Cl 3 Fe(OH)3 + Na. Cl

Double-Replacement Reactions • Volition only happen if i of the products – doesn't deliquesce in water and forms a solid – or is a gas that bubbles out. – or is a covalent compound usually water.

Your Turn Five • assume all of the reactions have identify. • Ca. Cl two + Na. OH • Cu. Cl 2 + K 2 S • KOH + Iron(NO 3)3 • (NH iv)2 And then 4 + Ba. F 2

Combustion Reactions • In a combustion reaction, a substance combines with oxygen, releasing a big amount of energy in the course of light and heat. • example: combustion of hydrogen 2 H 2(g) + O 2(g) 2 H two O(k) • case: combustion of propane C 3 H eight(g) + five O two(thou) 3 CO 2(g) + 4 H 2 O(g)

Combustion Reactions • Combustion • A compound composed of simply C H and maybe O is reacted with oxygen • If the combustion is complete, the products will be CO 2 and H 2 O. • If the combustion is incomplete, the products will be CO and H 2 O.

Your Turn VI • C iv H ten + O 2 (complete) • C 4 H 10 + O two (incomplete) • C half dozen H 12 O 6 + O ii (consummate) • C viii H 8 +O 2 (incomplete)

Your Turn Six Part II Distinguish between complete an incomplete combustion. Write a counterbalanced equation for the complete combustion of each of these compounds. a) acetic acrid, HC two H iii O two c) glycerol, C 3 H 8 O iii b) decane, C 10 H 22 d) sucrose, C 12 H 22 O 11 Write a balanced equation for the incomplete combustion of each of these compounds. a) glycerol, C 3 H 8 O three c) acetic acrid, HC 2 H 3 O 2 b) glucose, C six H 12 O vi d) acetylene, C ii H ii

How to recognize which blazon • • • Expect at the reactants A + B C Combination C A+B Decomposition C + AB CB + A Single replacement AB + CD Ad + CB Double replacement

Your Turn 7 • Determine the type of reaction and the products and balance. • H 2 + O 2 • H 2 O • Zn + H 2 SO 4 • Hg. O • KBr +Cl 2 • Ag. NO 3 + Na. Cl • Mg(OH)2 + H 2 And so 3

Permit's Review • Write out the balanced equation for Copper reacts with chlorine to form copper (II) chloride. • Write out the balanced equation for Zinc reacts with Hydrochloric acid to course Zinc chloride and hydrogen. • Write out the balanced equation for Calcium carbonate reacts with Zinc nitride to form Calcium nitride and Zinc carbonate.

Permit's Review • Balance the following equations – C seven H xvi + O 2 ---> CO ii + H 2 O – Na 2 O two + H 2 O ---> Na. OH + O ii – Al two(SO 4)3+Ca(OH)2 ----> Al(OH)iii+Ca. And then iv – NH 3 +HCl ----> NH four Cl – Ca 3(PO 4)two +Si. O 2 + C ----> Ca. Si. O 3 + CO + P

Permit'due south Review • What type of reaction are the following – – – Rb + S viii ---> Rb ii S NH 3 + O 2 ---> N 2 + H two O C + SO ii ---> CS 2 + CO C 3 H eleven + O two ---> CO 2 + H 2 O C 5 H 12 +O ii ---> CO + H 2 O Iron. Cl 3 + NH four OH --->Fe(OH)iii + NH 4 Cl

Let's Review • Balance them – – – Rb + S 8 ---> Rb 2 Southward NH three + O 2 ---> N 2 + H two O C + SO 2 ---> CS ii + CO C three H 11 + O 2 ---> CO two + H two O C 5 H 12 +O 2 ---> CO + H 2 O Fe. Cl iii + NH 4 OH --->Fe(OH)3 + NH 4 Cl

Allow'southward Review • Predict the products and balance – Sodium phosphate reacts with Lithium metal. – Butane (C 4 H 10) is completely combusted. – Sodium Chloride reacts with Silver Hydroxide – Aluminum Bromate reacts with Copper – Potassium reacts with Sulfur – Octane (C 8 H xviii) is incompletely combusted

Let'southward Review • Predict the products and balance – – – – H 2 + O 2 Al 2 Te 3 Zn + H 2 SO 4 Hg. O KBr +Cl ii Ag. NO 3 + Na. Cl Mg(OH)2 + H 2 Then three

Let'south Review • Using the table on Pg 286. Predict if these reactions will occur – – – – Li + Ba. South Fe + KCl Au + Hg. S Mn + Cu. S Ba + Li. Southward Na + KCl HCl + Pb Pb. Cl 2 + H two
Source: https://slidetodoc.com/chemical-equations-and-reactions-chapter-8-describing-chemical/
Belum ada Komentar untuk "Chemical Equations and Reactions Chapter 8 Review Answer Key"
Posting Komentar